Yes, you can overboil protein samples prepared in Laemmli buffer with β-mercaptoethanol (BME), leading to potential degradation, aggregation, or smearing on SDS-PAGE gels. Standard boiling is 95–100°C for 3–5 minutes; exceeding this (e.g., 8+ minutes) risks poor band resolution, especially for heat-sensitive or large proteins.
Why Boiling Is Used
Boiling destroys proteins, exposes their structure to SDS and promotes disulfide bond reduction by BME (an important Laemmli buffer constituent). This allows the correct separation of samples with regard to molecular weight in Western blotting or SDS-PAGE. These effects are provided by the presence of SDS, glycerol, bromophenol blue, Tris-HCl, and BME (or DTT) in Laemmli buffer (which is usually 4X stock, diluted to 1X).
What Happens If You Overboil?
- Degradation/Aggregation: Prolonged heat (>5–8 minutes) can cause irreversible damage, forming aggregates that appear as smears or faint bands instead of sharp ones.
- Loss of Protein Integrity: Heat-sensitive proteins (e.g., membrane or large complexes) may precipitate or lose epitopes, reducing detection in blots.
- Examples from Labs: Users report no bands after 8-minute boils or thick smears after 1+ hour exposure; re-boiling stored samples is generally safe but not ideal.
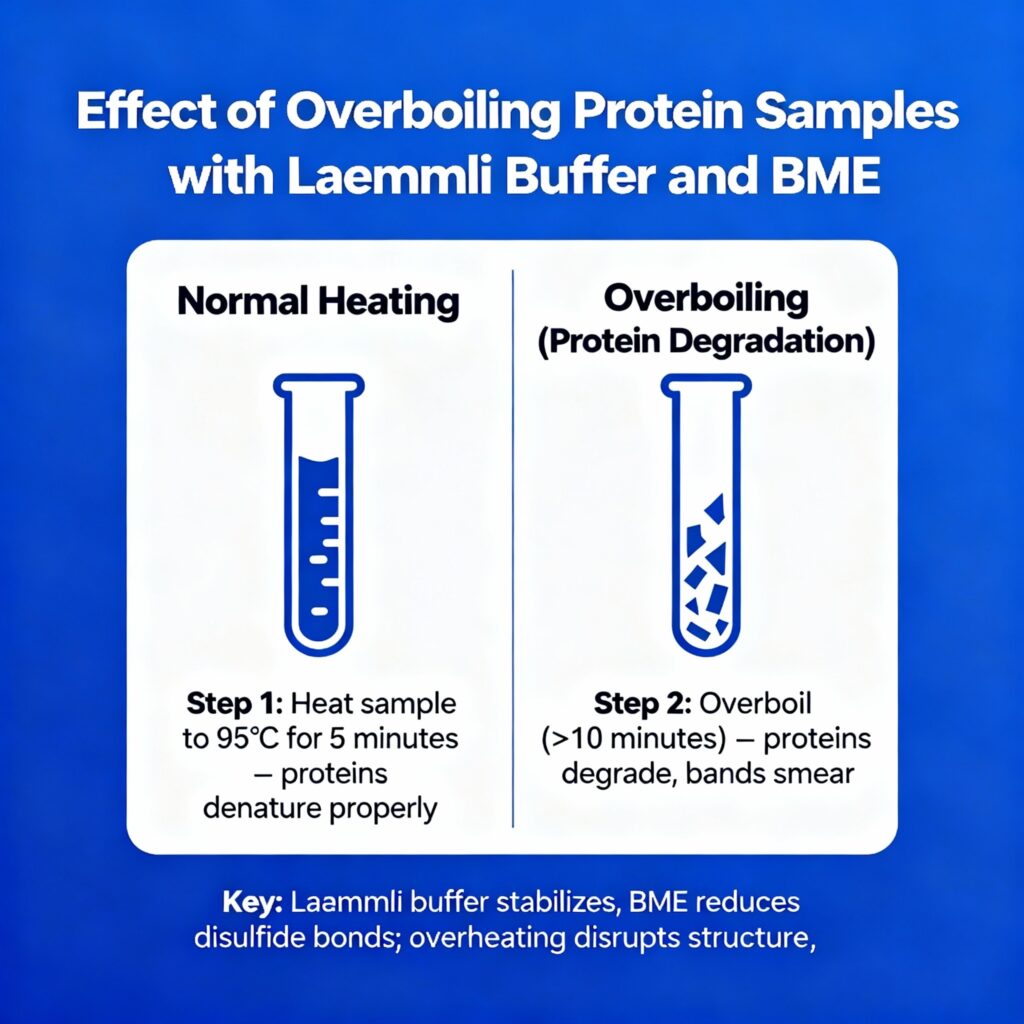
Best Practices to Avoid Overboiling
- Optimal Conditions: Boil at 95–100°C for 3–5 minutes, then briefly centrifuge to remove condensation.
- For Sensitive Proteins: Skip boiling or use 37–70°C for 10–30 minutes; test empirically.
- Buffer Tips: Add fresh BME/DTT just before boiling (it’s unstable); store aliquots at -20°C.
- Recovery: If overboiled, dilute and re-run; freezing post-boil is fine for short-term storage.
If your protein fails to appear, verify loading controls (e.g., β-actin) and adjust conditions per protein type. For protocols, see Laemmli’s original 1970 paper.
Last modified: October 14, 2025